
Transparency
Transparency
SIRO offers a range of services, from clinical operations to data services, data analytics and medical
writing in compliance with international standards. Get in touch for more information or queries. We
would be delighted to be of any assistance.
Home » SERVICES » Medical Writing » Transparency
List of Services
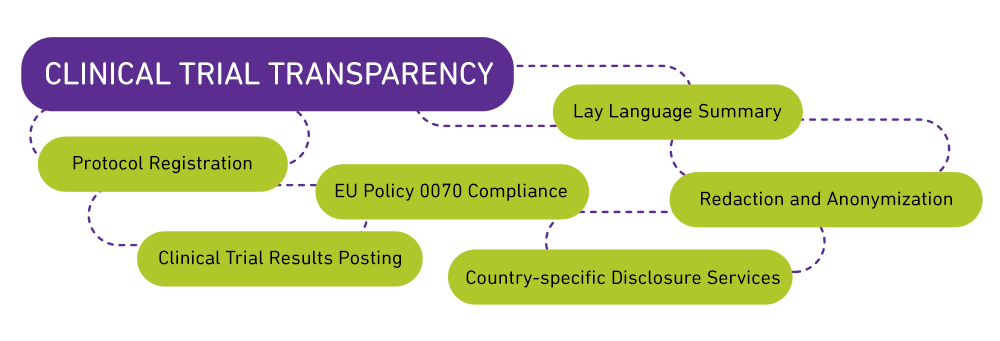
Sub-services

Protocol Registration
- PRF for ClinicalTrials.gov
- CTA for EudraCT
- Protocol annual and rapid updates
- Country-specific protocol registrations
Results Posting
- RRF and ERF authoring
- PRS/NIH Comments addressal
- Standalone QC of RRF, ERF
- With or without XML files
- Results posting through PharmaCM, vCTR or directly on ClinicalTrials.gov or EudraCT portal
- Country specific registries Eg: Germany (DRKS), Spain (REEC), Australia and New Zealand (ANZCTR), India (CTRI) – With translation into local languages


Redaction and Anonymization
- Compliance to EU Policy 0070
- Redaction of clinical reports to protect personally identifiable information (PII), protected-personal data (PPD) and company confidential information (CCI)
- De-identifying and masking personal data and commercially confidential information
- Risk assessment
- Anonymization report
- Randomization to anonymize or de-identify PII or protected health information (PHI)
- Adherence to TransCelerate Guidelines
Lay (Plain) Language Summaries
- Writing of lay summaries
- Standalone QC
- Translation into local languages


